The opioid epidemic has led medical professionals and patients to explore alternatives to potentially addictive pharmaceuticals, such as light therapy.
Photobiomodulation (PBM) therapy, a form of light therapy using red and near-infrared light to stimulate cell growth and reduce inflammation, is gradually gaining acceptance by the medical community. The noninvasive technology, discovered in 1967, is approved by the U.S. Food and Drug Administration (FDA) to manage pain and treat a variety of medical conditions.
Patients receive therapy several times a week for several weeks before they see pain relief.
PBM has been used in over 100 million patient treatments without documented side effects, according to the PBM Foundation, and has been shown to reduce the cost of care by 73 percent. It has been supported by over 940 randomized clinical trials and 9,000 research studies and was the focus of a Congressional Briefing on October 11, 2018, a White House briefing on April 9, 2019, and meetings with several federal health agencies
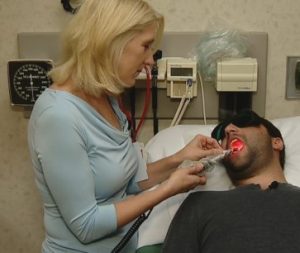
(Photo courtesy of the PBM Foundation: Oncology Care Manager Annette Quinn treating a patient with PBM to reduce the side effects of cancer chemotherapy at the University of Pittsburgh Medical Center (UPMC).
Drug-Free Pain Relief
PBM is usually an alternative to addictive pain medications, says Gerry Ross, D.D.S.
“I have been using PBM in my practice for 30 years, and in that time, I never wrote prescriptions for post-op opioid narcotic prescriptions,” said Ross. “In 99 percent of the cases, patients need nothing [for pain] or use over-the-counter pain meds.”
PBM can be used after complicated procedures, says Ross.
“I do surgical extractions, impactions, and periodontal-flap surgery,” said Ross. “In addition, I have been treating facial pain, TMD (temporomandibular joint disorders), and trigeminal neuralgia on a referral basis. Not only do patients not need pain meds, [they] don’t require muscle relaxants, anti-inflammatory drugs, or amitriptyline.”
Other professionals have adopted PBM technology after taking one of the more than 200 courses he has taught, says Ross.
“I recently taught an oral surgeon who travels to different general practices doing impactions and difficult surgical cases and she immediately stopped needing to prescribe opioids,” said Ross.
Therapeutic Limits
Multiple studies have shown PBM light therapy is beneficial above a certain dose threshold but less effective or even damaging above a higher threshold, which is called a biphasic dose-response.
Currently, PBM requires close monitoring of exposure levels, careful application to deliver precise dosages and wavelengths for the particular condition treated, and extensive training for clinical technicians, says Ross.
“The World Association for Photobiomodulation Therapy, formed in 1994 in Barcelona, is working on this and is looking to publish parameters for all treatments,” said Ross. “There are numerous different companies using different wavelengths and many will make claims to sell units that are not based on the science. “
There are currently more than 4,000 published research papers examining the healing power of PBM, some of which have found PBM therapy is not very effective, says Ross.
“Some of the conflicting research may be because PBM eliminates the use of many pharmaceuticals, with much of the research being sponsored by the companies that manufacture these drugs, while many PBM companies do not have the funds to back their own research,” said Ross.
Treats ‘Long Covid’
Earlier this year, Shepherd University (SU) in Shepherdstown, West Virginia opened a PBM light therapy center using a $500,000 grant under the Coronavirus Aid, Relief, and Economic Security (CARES) Act. The center will “…focus on wellness, explore deaddiction to opioids, and treat long COVID-19 symptoms,” says Parveen Arany, M.D, the center’s interim executive director, according to an SU press release on March 15.
“A lot of people have had COVID-19 and as they are recovering, we are finding things like fatigue, depression, [and] other kinds of chronic diseases that are causing concern,” said Arany. “We would like to use this innovative treatment that is nonpharmacological and noninvasive and focuses on the host’s resilience. We are trying to make people healthier and better.”
‘Antiquated FDA Approval Process’
The spread of therapies has been slowed by the sluggish FDA approval process for therapies and devices, says Edward Hudgins, Ph.D., founder of the Human Achievement Alliance.
“PBM will be further developed and new applications created to the extent that medical research entrepreneurs are free to innovate,” said Hudgins. “This freedom will depend in part on reducing government regulations and certification requirements.
“A recent Tufts analysis found that on average it takes 10 to 12 years at a cost of $3 billion to bring a new medication from research lab to market,” said Hudgins. “This is in large part because of an antiquated FDA approval process.”
‘Patients Suffer Needlessly’
Medical progress has been slowed by limiting access to potentially beneficial treatments, says Jeffrey Singer, M.D., a senior fellow at Cato Institute.
“Unfortunately, the FDA regulatory system has thrown a lot of sand in the gears of the discovery process, often making patients suffer needlessly for years before being allowed to try the treatment or device,” said Singer.
“As a matter of principle, I think it’s always a good thing to discover new, effective ways of relieving pain that are noninvasive and have fewer side effects, including drug dependence,” said Singer. “And it’s also important to keep in mind that a therapeutic method that works for some people may not work for others,” said Singer. “There are many factors that go into how an individual patient responds to a specific drug or therapeutic treatment.”
Kevin Stone (kevin.s.stone@gmail.com) writes from Arlington, Texas
Internet info:
“A Non-Drug, Non-Invasive Treatment for Pain – Light Waves (Guest: Scot Faulkner), The Heartland Daily Podcast, April 20, 2022:
Photo courtesy: PBM Foundation
This article was updated on May 29, 2022.
For more great content from Health Care News.
For more information:
https://pbmfoundation.org
Clinical trials:
https://clinicaltrials.gov/ct2/results?cond=&term=Photobiomodulation&cntry=&state=&city=&dist



















